DESCRIPTION
SPECIFICATIONS
INFO / VIDEO
RELATED PRODUCTS
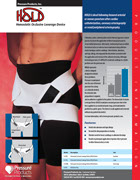
Maintains pressure over femoral arterial or venous puncture site after cardiac catheterization, coronary arteriography or renal/peripheral arteriography.
Following cardiac catheterization via the femoral approach current practice has been the application of direct manual pressure to secure initial hemostasis. Afterwards, continued pressure on either the femoral artery or vein has been maintained using adhesive-elastic bandages and/or sandbags.
Skin irritation, abrasions, and tape allergy, all compound the discomfort associated with the application and removal of the adhesive dressing. Although increasing pressure, it is difficult to maintain sandbags in a stable position over the puncture site.
HOLD represents a device uniquely designed to obviate these problems. Pressure is maintained over the area of femoral puncture for as long as is clinically desired. No extensive skin preparation is required, and no adhesive is applied to the patient.
The Hemostatic Occlusive Leverage Device (HOLD) maintains constant pressure due to the force applied via an elasticized groin strap, and maintained in position by a pelvic apron. The device is easily applied and removed without any patient discomfort.
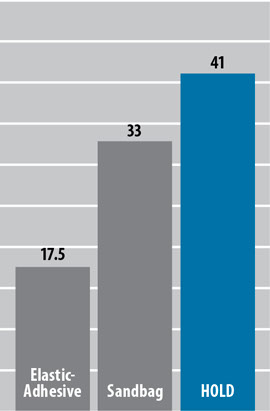
Femoral Pressure Comparison
(mm Hg)
HOLD is a trademark of Pressure Products Medical Supplies, Inc..
HEMOSTATIC OCCLUSIVE LEVERAGE DEVICE (HOLD)
Model
Size
H-100S
Small
H-150L
Large
H-200XL
Extra Large
Features
![]() Avoids risk of skin abrasion and tape allergies
Avoids risk of skin abrasion and tape allergies
![]() No need for skin preparation or shaving
No need for skin preparation or shaving
![]() Obviates need for heavy sandbags or adhesives
Obviates need for heavy sandbags or adhesives
![]() Results in prolonged application of greater direct pressure to either femoral artery or vein
Results in prolonged application of greater direct pressure to either femoral artery or vein

SAFESHEATH INTRODUCERS AND SEALING ADAPTERS
SAFESHEATH CS AND LV INTRODUCERS
RIGHT-SIDED CS / RV ACCESS INTRODUCER
CORONARY SINUS VENOGRAPHY BALLOONS
ACCESSORIES
TRANSSEPTAL ACCESS
HEMOSTASIS & COMPRESSION MANAGEMENT
PACEMAKER PROTECTION AND COMFORT
Search Pressure Products
PRODUCTS
PROCEDURES
EDUCATION
ORDER
Pressure Products offers a complete line of vascular access and lead delivery systems and accessories, as well as medical devices for femoral and pacemaker pocket hemostasis.
Copyright © 2022 Pressure Products Medical Supplies, Inc. All rights reserved.